 This greenery then “exhales” oxygen, releasing it back for animals and other organisms to use. (Many microbes do, too.) Plants “breathe in” this CO 2 and use it for photosynthesis. A warm, life-sustaining jacketĪnimals - be they humans, whales, turtles, fish or insects - “exhale” CO 2 when they breathe. But since fire cannot burn without oxygen, CO 2 is used in some fire extinguishers as a quick, effective way to douse flames. In some cases, this can cause a person - or other animal - to suffocate. If CO 2 builds up rapidly, such as at the bottom of a tunnel or in an enclosed room, this gas will push oxygen out of its way. It’s also the source of the fizz in carbonated drinks.ĬO 2 is heavier than air, which means it will sink. (Always be careful around dry ice! It can cause frostbite quickly.)ĬO 2 can be captured in high-pressure canisters and used to inflate bicycle tires or power paintball guns. If you’ve ever walked through a haunted house with a so-called “smoke” machine, the fog is likely vapor from a block of dry ice. This “dry ice” looks snowy white and can transform directly from a solid to gas. But cool it to -78° Celsius (-109° Fahrenheit) and it turns solid. “But with three atoms all in a line, CO 2 can wiggle the oxygens at each end.” This helps the molecule absorb heat when the sun’s rays hit it.Īt typical conditions on Earth, CO 2 is colorless and odorless. “Molecules with just two atoms, like oxygen (O 2) and nitrogen (N 2), really can’t wiggle much,” he says. He describes CO 2 molecules as being able to “flap their wings” a little bit. Mark Potosnak is an Earth scientist at DePaul University in Chicago, Ill. (The CO 2 molecule is also nonpolar, which means that all of the electric charges inside it are spread out relatively evenly.) This means that the carbon shares two pairs of electrons - four electrons in all - with each of the oxygen atoms. One reason it forms this straight-line shape is its two double covalent (Koh-VAY-lunt) bonds. 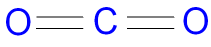
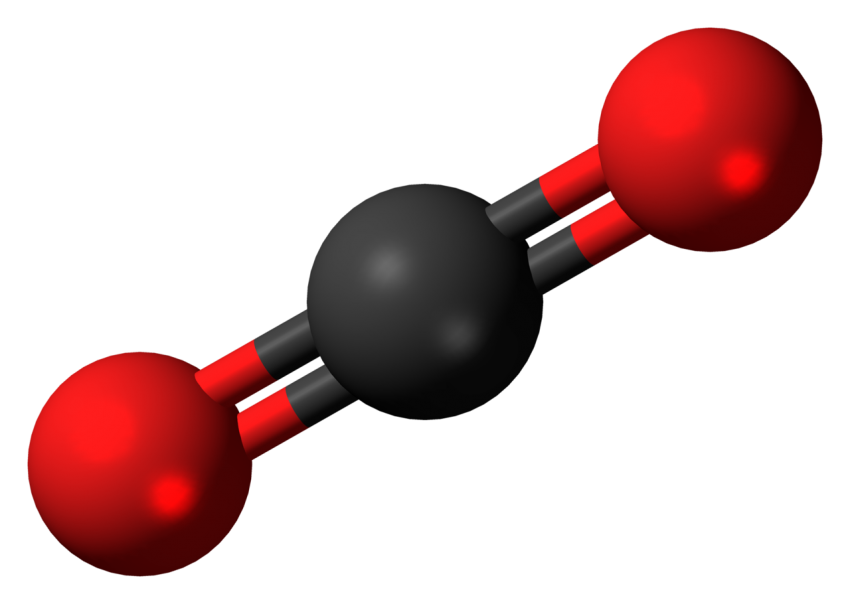
If you could zoom in to see a single molecule of CO 2, it would look like a straight line with a carbon atom in the center and oxygen atoms at either end. Science Photo Library/Science Photo Library/Getty Images The gray attachments show the double bonds that give this molecule its straight-line structure. All rights reserved.This illustration of a molecule of carbon dioxide shows a central carbon atom (black) linked to oxygen atoms (red) at either end. On behalf of the United States of America. Shall not be liable for any damage that may result fromįor NIST Standard Reference Data products. However, NIST makes no warranties to that effect, and NIST /carbondioxide-56a128af3df78cf77267ef26.jpg)
Uses its best efforts to deliver a high quality copy of theĭatabase and to verify that the data contained therein haveīeen selected on the basis of sound scientific judgment. The National Institute of Standards and Technology (NIST)
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
